Source: News 12 NJ
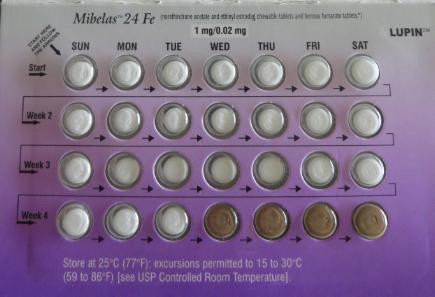
EDISON – Lupin Pharmaceuticals Inc. has issued a voluntary nationwide recall of its Mibelas 24 Fe products due to a labeling mistake that could result in unintended pregnancies, according to the U.S. Food and Drug Administration.
A confirmed market complaint indicated a packaging error, where the blister was rotated 180 degrees within the wallet, reversing the weekly tablet orientation and making the lot number and expiration date no longer visible. The first four days of therapy would have had four non-hormonal placebo tablets as opposed to the active tablets.
As a result of the packaging error, oral contraceptive tablets that are taken out of sequence may place the user at risk for contraceptive failure and unintended pregnancy.
The company said reversing of the order may not be apparent to either new users or previous users of the product, increasing the likelihood of taking the tablets out of order.
Consumers who have the affected product should notify their physician and return the product to the pharmacy or place of purchase.
Consumers with questions regarding this recall can contact Lupin by phone at 1-800-399-2561, between 8 a.m. and 5 p.m. EST, Monday through Friday. Consumers should contact their physician or healthcare provider if they have experienced any problems that may be related to taking the product.